OUR RESEARCH
Our lab is harnessing the power of systems biology methods – including high-throughput single cell sequencing technologies, combinatorial perturbations, and innovative computational tools – to understand the context-dependent role of inflammatory signaling in metastasis. We explore tumor cell extrinsic and intrinsic ways in which metastases adapt resistance to this chronic stimulus and its impact on bi-directional crosstalk between tumor cells and their microenvironment.
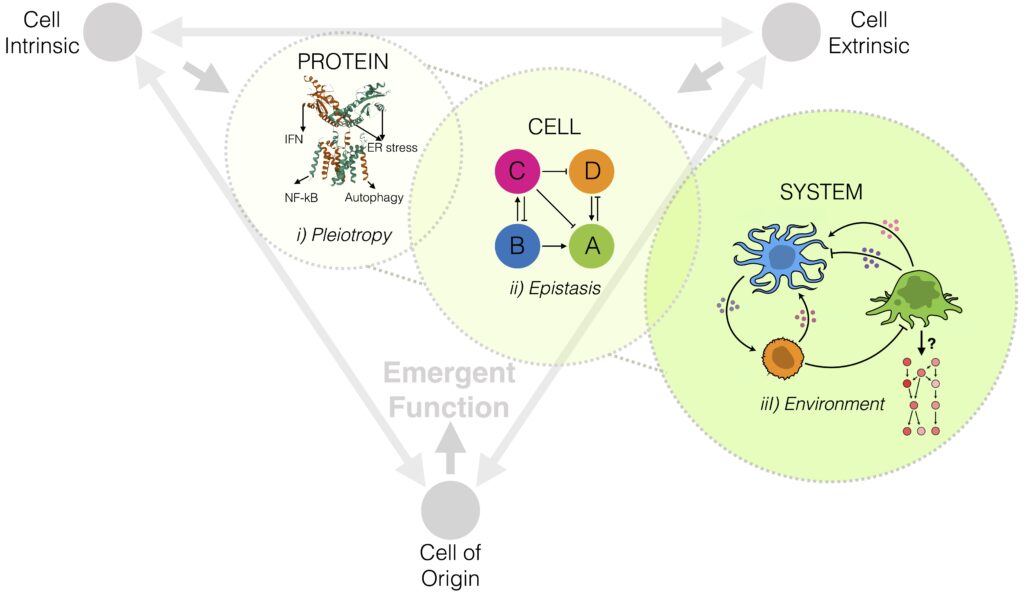
The genotype-to-phenotype problem. Determining how genome-encoded components perform cellular function is complicated by the pleiotropy found in biological systems. Pleiotropy refers to when a single gene influences multiple unrelated traits. As such, perturbations to a single gene can affect multiple, independent cellular responses – complicating our ability to infer sequence-function relationships. This problem, otherwise known as genotype-to-phenotype mapping, represents one of biology’s most intractable challenges. Combining high–throughput single cell sequencing with development of innovative computational tools, synthetic biology approaches and mouse models of cancer, our lab asks how the same genome-encoded components can give rise to diverse phenotypes, or cellular functions, at different stages of disease progression. We focus on cancer metastasis as our model of a complex, evolutionary process and develop novel approaches that tackle fundamental problems encountered in genotype-to-phenotype mapping, including: 1) Pleiotropy, 2) Epistasis and 3) Microenvironmental Crosstalk.
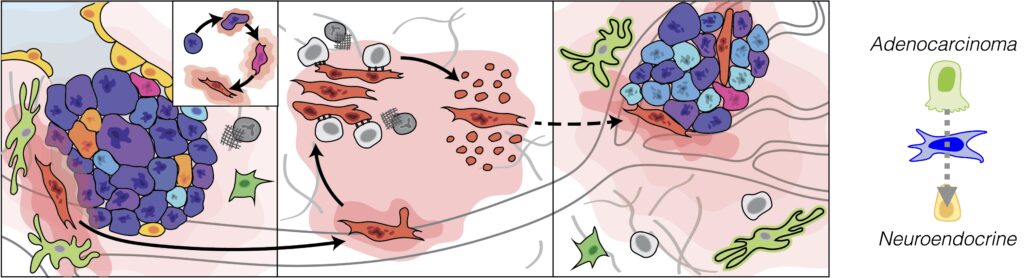
Cancer metastasis as a complex, multicellular process. Most cancer deaths are caused by metastasis. The invasion-metastasis cascade releases large numbers of disseminated tumor cells throughout the body, even during the early stages of tumor growth. A minority survive as potential seeds for future metastatic outbreaks. They must adapt to a less hospitable microenvironment, evade immune surveillance and progress from the micro- to macro-metastatic stage, resulting in generation of a secondary tumor. All of these processes trigger inflammation and are critically regulated by cellular microenvironmental crosstalk, events central to a wound-healing response. We are inspired by the following recent discoveries:
- The presence of nucleic acids in the cytosol is a critical source of chronic inflammatory signaling emanating from tumor cells that promotes metastasis. Read more.
- Tumor stemness, which varies with emergence from the micro- to macro-metastatic stage, is intimately coupled to inflammatory response programs that engender distinct susceptibilities to innate immune cell killing. Read more.
One way in which cells adapt diverse functional responses is through context-dependent interactions with their microenvironment. Cells typically send messages to their microenvironment by emitting ligands, which bind to complimentary receptors on the surfaced of target cells; then triggering a change in the behavior of the target cell. Complimentary ligand-receptor-pairs are well annotated and methods have been developed to infer cell-cell interactions from single cell expression data based on their mutual expression. However, existing work views each cell as an independent unit. We have taken the alternative view that cells sampled from the same tumor microenvironment are fundamentally participating in an underlying biological process and are working towards generating a principled understanding of how cells coordinate their behavior; which has the potential unlock deep biological insights into cancer progression, and suggest new targets for effective treatments. Exploiting inter- and intra-sample variability, we are developing new methods to infer the effect of a ligand-receptor-mediated interaction on the tumor microenvironment and to characterize intercellular network dynamics.